Leave Your Message
The compound 8 Hydroxy 5 Nitroquinoline is gaining attention in various fields. Its unique chemical structure offers potential benefits in multiple applications. Research is exploring its role in medicinal and analytical chemistry.
One of the key areas of focus is 8 Hydroxy 5 Nitroquinoline applications in drug development. This compound has shown promise in providing therapeutic effects against certain diseases. Additionally, its use in chemical analysis has emerged as a valuable tool for researchers. The compound's ability to interact with specific biological targets broadens its application scope.
However, there are challenges to overcome. The complexity of its synthesis can deter experimentation. Moreover, potential toxicological effects must be studied extensively. Understanding these aspects is crucial for realizing its full potential in practical applications. As the research progresses, refining methodologies is necessary for better utilization. The journey of exploring 8 Hydroxy 5 Nitroquinoline applications is just beginning.
8 Hydroxy 5 Nitroquinoline is a chemical compound that attracts interest in various fields. It is mainly studied for its potential applications in medicinal chemistry. This compound has unique properties that may offer therapeutic benefits.
The chemical structure of 8 Hydroxy 5 Nitroquinoline enables it to interact with biological systems. Researchers investigate its role in inhibiting specific enzymes. Additionally, its potential as an antibacterial agent is a subject of ongoing studies. These features highlight its importance in drug development.
However, understanding 8 Hydroxy 5 Nitroquinoline is not without challenges. The synthesis process can be complex and requires careful handling. Researchers must navigate safety protocols to avoid potential hazards. Continuous research is necessary to unlock its full potential for practical applications. Exploring its capabilities may lead to significant advances in health and science.
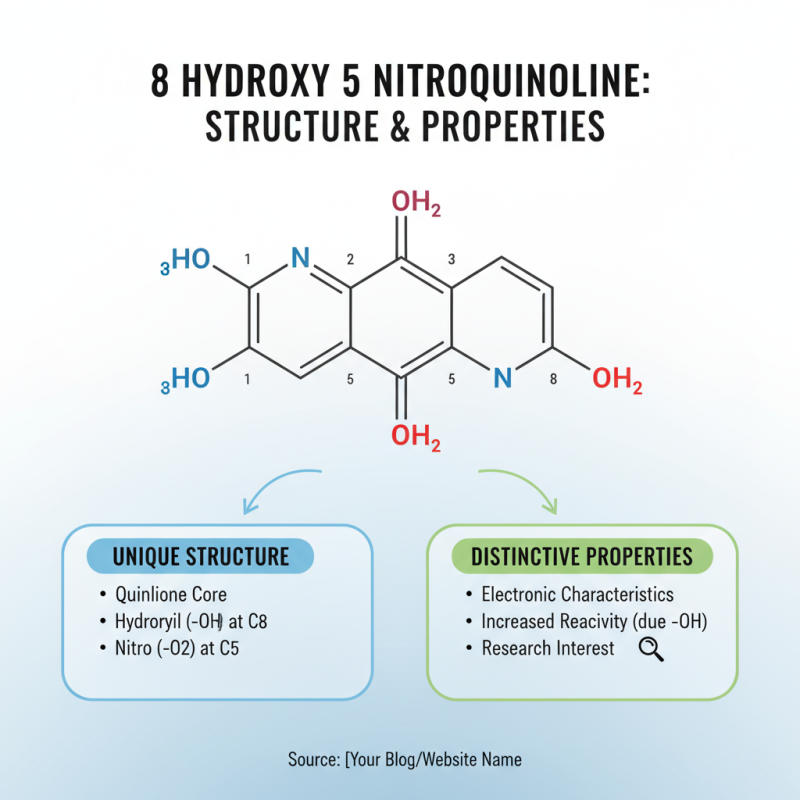
8 Hydroxy 5 Nitroquinoline is an intriguing compound, noteworthy for its unique chemical structure. The core structure consists of a quinoline ring that is modified by hydroxy and nitro groups. This arrangement leads to distinctive electronic characteristics. The presence of the hydroxy group contributes to its reactivity, making it an interesting subject for study in various chemical reactions.
The compound exhibits specific properties that enhance its applications. For instance, its solubility in organic solvents allows it to be utilized in diverse chemical environments. The nitro group introduces additional reactivity, which can be beneficial in synthetic pathways. Researchers often explore these properties for applications in dyes and pigments. This compound can also serve as an important intermediate in pharmaceuticals and agricultural chemicals.
While 8 Hydroxy 5 Nitroquinoline shows great promise, there are challenges in its application. The stability of the compound under various conditions raises questions. It is essential to consider these aspects when designing experiments or products. Understanding the balance between its advantageous properties and potential limitations can lead to more effective usage in the future.
The synthesis of 8 Hydroxy 5 Nitroquinoline involves several important methods. One common approach is the nitration of quinoline, followed by hydroxylation. This two-step process allows for the introduction of both the nitro and hydroxy groups. The conditions of the reaction, such as temperature and reagents, significantly affect the yield and purity of the product.
Another method involves the use of various catalysts to enhance the reaction efficiency. This can include transition metal catalysts that facilitate the nitration step. Experimentation with different catalysts can lead to improved reaction rates and reduced by-products.
Tips: Always monitor reaction parameters closely. Controlling temperature can prevent unwanted side reactions. Conduct small-scale trials to optimize conditions before scaling up. Understanding the limitations of your methods is crucial. The synthesis can be challenging, and sometimes the desired product may not form as expected. Adjusting your approach based on practice and feedback is essential for success.
| Property | Details |
|---|---|
| Chemical Formula | C9H6N2O3 |
| Molecular Weight | 178.16 g/mol |
| Synthesis Method | Electrophilic nitration followed by hydroxy substitution |
| Solubility | Soluble in organic solvents, such as ethanol and acetone |
| Uses | Used in pharmaceuticals, dyeing, and as an analytical reagent |
| Toxicity | Considered hazardous; proper safety precautions needed |
8 Hydroxy 5 Nitroquinoline has garnered attention in medicinal chemistry. Its unique chemical structure contributes to diverse applications in drug development. The compound features a nitro group and hydroxyl group, enhancing its biological activity.
In medicinal chemistry, researchers investigate its potential as an antibacterial agent. Preliminary studies show promising results against various bacterial strains. The compound’s ability to inhibit certain enzymes suggests it may disrupt bacterial growth. Additional research is necessary to fully understand its mechanisms.
Beyond antibacterial properties, 8 Hydroxy 5 Nitroquinoline might offer insights for cancer treatment. Some studies indicate it could target cancer cells effectively. However, the compound's safety profile remains unclear, requiring extensive evaluations. Preparing for potential side effects is crucial as scientists push forward with clinical trials. This ongoing exploration challenges researchers to refine their understanding and application of the compound.
Research on 8 Hydroxy 5 Nitroquinoline (8-HNQ) reveals promising potential across various fields. This compound is notable for its applications in biochemistry and medicinal chemistry. Its unique structure allows it to interact with biological targets effectively. However, there are several challenges researchers face.
One challenge is the compound's stability. 8-HNQ can degrade under certain conditions, limiting its shelf life and usability. Researchers need to explore formulations that protect its integrity. Another issue is the limited understanding of its long-term effects on human health. Studies are still ongoing to establish safety profiles for various applications.
Tips: Maintaining stringent lab protocols is vital for preserving compound integrity. Ensure that all experiments are conducted in controlled environments to prevent degradation.
As research advances, new applications may emerge, but funding remains a concern. Many projects lack adequate support, slowing progress. Researchers should seek collaborations to pool resources and knowledge, addressing the funding gap.
Tips: Networking in the scientific community can open doors to collaborative funding opportunities. Attend conferences and workshops to build connections that can lead to fruitful partnerships.